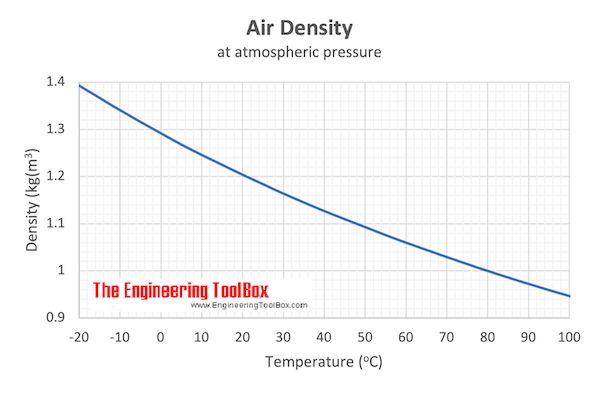
If the specific heat capacity of air at constant pressure is 993 J kg ^(-1) K ^(-1) calculate specific heat capacity at constant volume ? Density of air at N.T.P. is 1.293 Kg //m ^(3). [E.Q.)

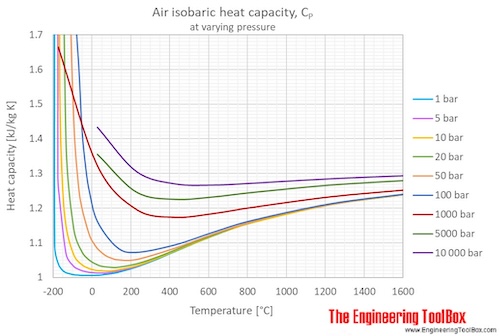
From given data, calculate the value of the mechanical equivalent of heat. The specific heat capacity of air at constant volume is 170 cal/kg - K, gamma = CP/CV = 1.4 and

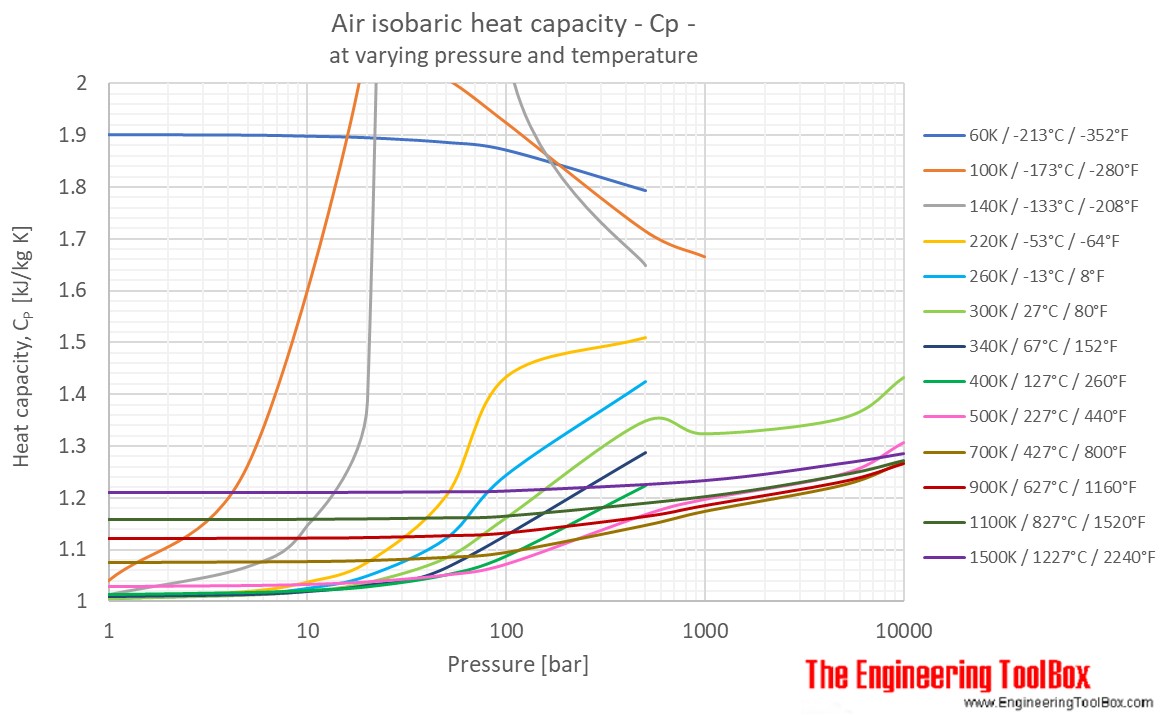
3: Specific heat at constant pressure vs. temperature, for air and CO 2 . | Download Scientific Diagram

The specific heat of air at constant pressure is `1.005 kJ//kg//K` and the specific heat of air at - YouTube

Variation in specific heat at constant pressure with temperature for... | Download Scientific Diagram

The specific heat of air at constant pressure is 1.005 kJ/kg K and the specific heat of air at constant volume is 0.718 kJ/kg K .Find the specific gas constant.

The specific heat of air at constant pressure is 1.005 kJ/kg/K and the specific heat of air at constant volume is 0.718 kg kJ/kg/k . If the universal gas constant is 8.314

SOLVED: of '390 K The density of A hot air balloon contains 850 cm' ofair at an assumed constant temperature the air at this temperature is 0.903 m J heat of the