How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrop

Molecules | Free Full-Text | How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrophilicity Indices
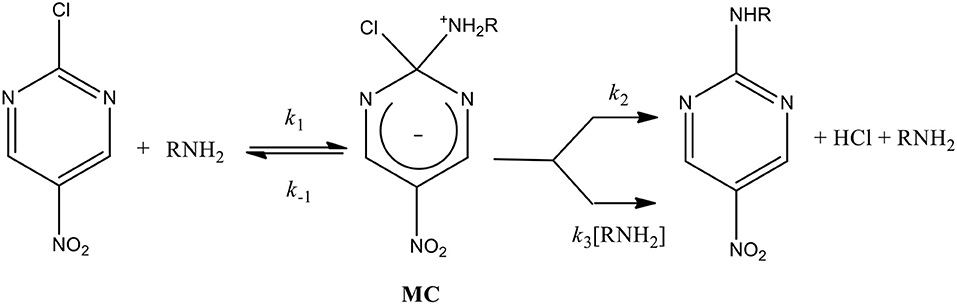
Frontiers | Experimental Analyses Emphasize the Stability of the Meisenheimer Complex in a SNAr Reaction Toward Trends in Reaction Pathways

Secreted reporter assay enables quantitative and longitudinal monitoring of neuronal activity | bioRxiv

Evidences for the key role of hydrogen bonds in nucleophilic aromatic substitution reactions. | Semantic Scholar

Unusual Spectroscopic and Electric Field Sensitivity of Chromophores with Short Hydrogen Bonds: GFP and PYP as Model Systems | The Journal of Physical Chemistry B

Sulfite-Catalyzed Nucleophilic Substitution Reactions with Thiamin and Analogous Pyrimidine Donors Proceed via an SNAE Mechanism | The Journal of Organic Chemistry

Meisenheimer complexes as hidden intermediates in the aza-S N Ar mechanism - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB00600A
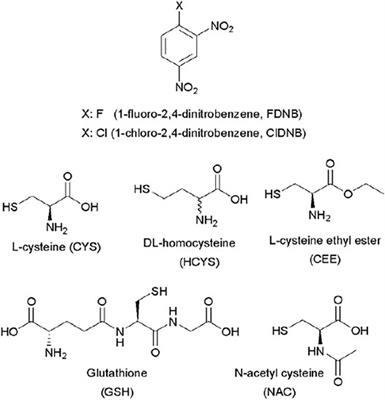
Frontiers | Kinetics and Reaction Mechanism of Biothiols Involved in SNAr Reactions: An Experimental Study
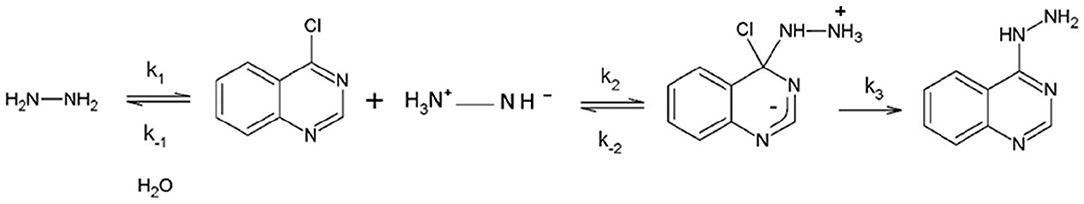
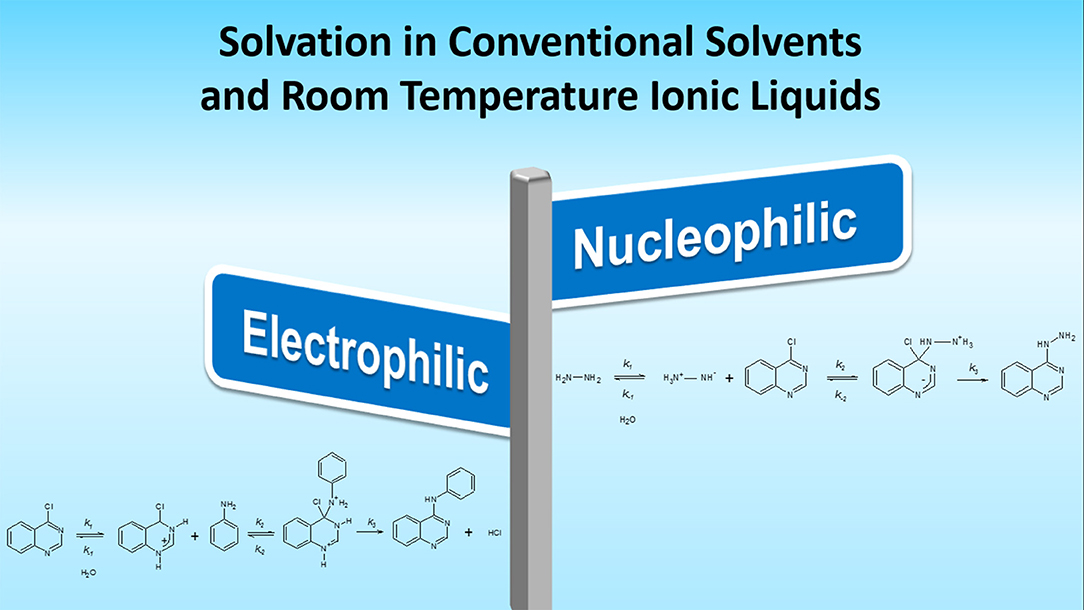
Frontiers | Activation of Electrophile/Nucleophile Pair by a Nucleophilic and Electrophilic Solvation in a SNAr Reaction
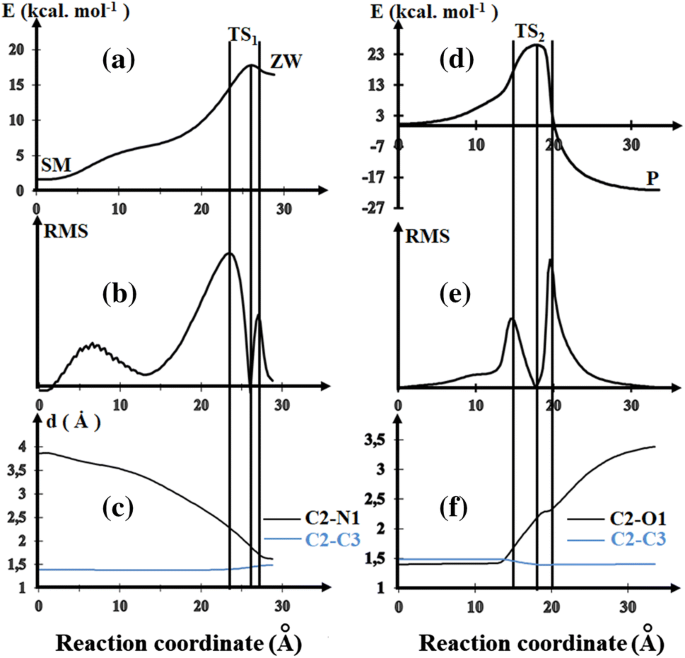
First-principles study of the reaction mechanism governing the SNAr of the dimethylamine on 2-methoxy-5-nitrothiophenes | SpringerLink
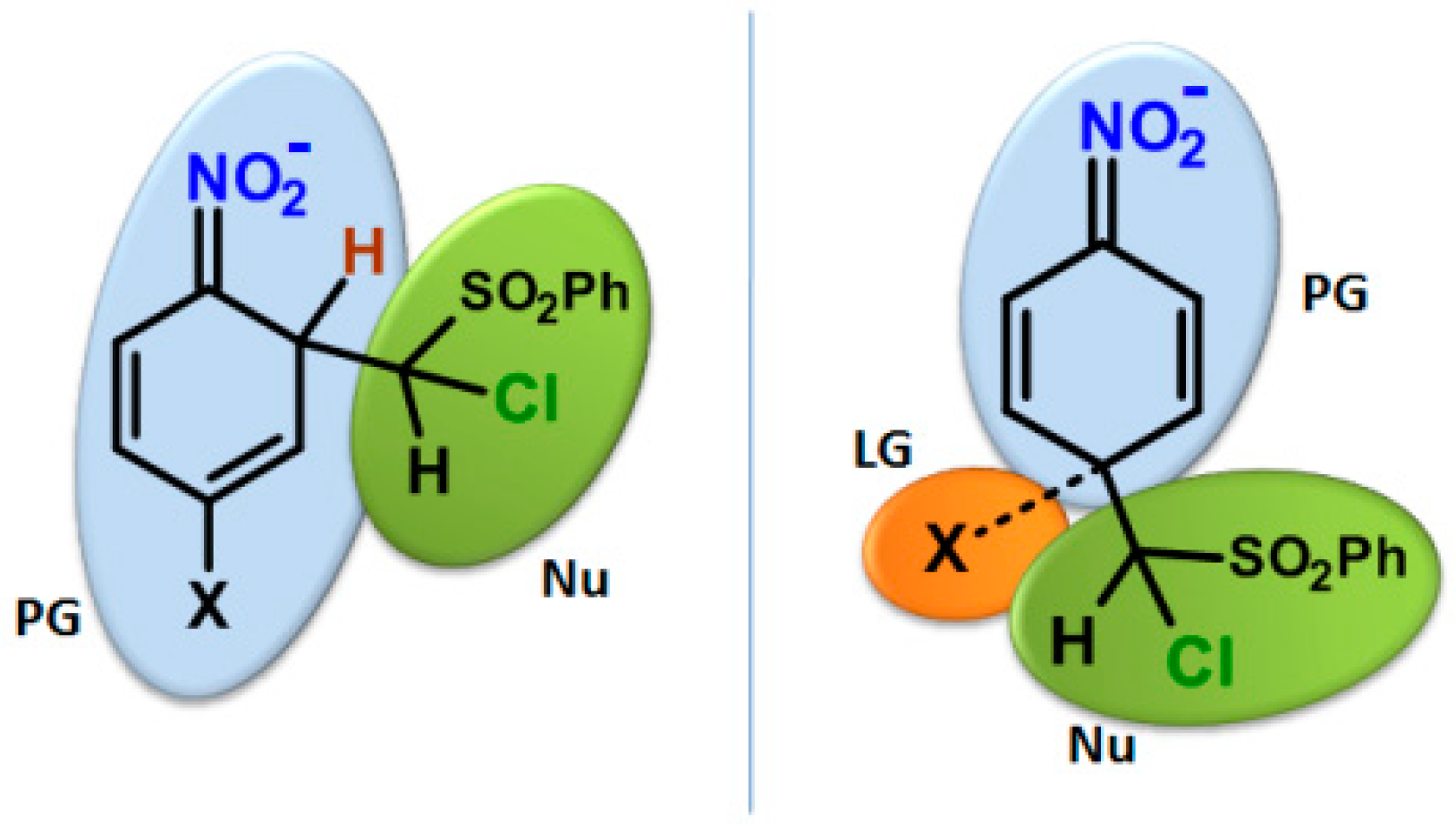
Molecules | Free Full-Text | How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrophilicity Indices
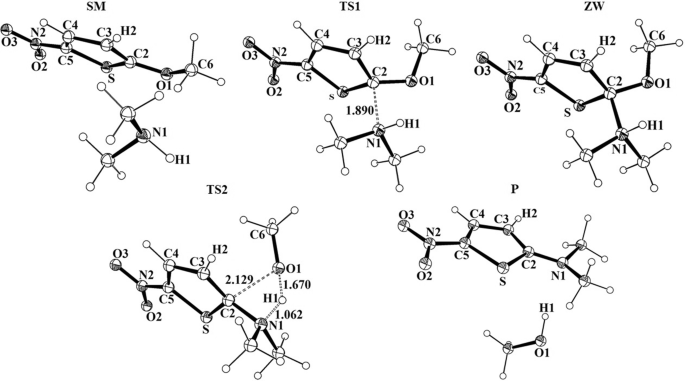
First-principles study of the reaction mechanism governing the SNAr of the dimethylamine on 2-methoxy-5-nitrothiophenes | SpringerLink

Potential energy profiles for the limiting step of S N Ar-H (left) and... | Download Scientific Diagram

Molecules | Free Full-Text | How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrophilicity Indices