How to Balance P4 + Cl2 = PCl3|Chemical eqations P4+Cl2=PCl3| Chemical Reaction Balance P4+Cl2=PCl3 - YouTube
✓ Solved: Phosphorus pentachloride gives an equilibrium mixture of PCl5, PCl3, and Cl2 when heated. PCl5(g)⇌...

One litre of mixture of CO and CO2 is passed through red hot charcoal in tube. The new volume becomes 1.4 litre . Find out
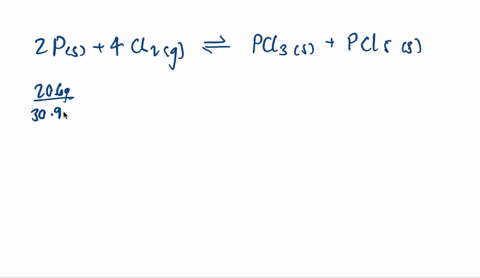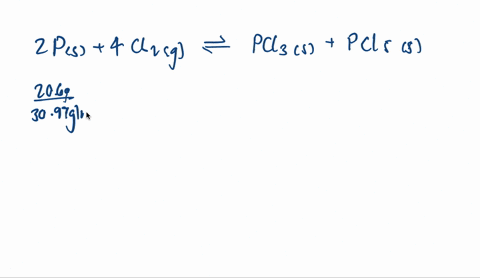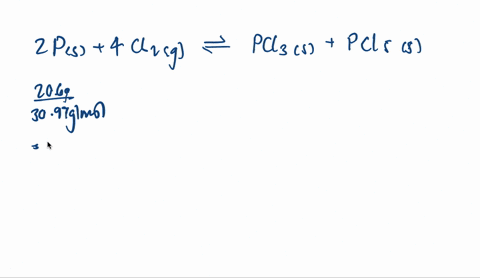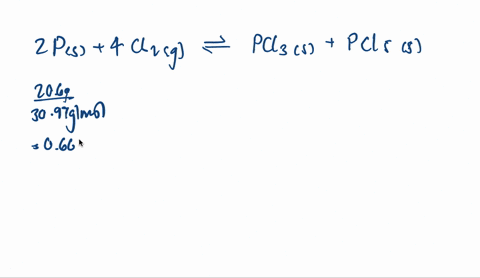
SOLVED:A mixture of 20.6 g of P and 79.4 g Cl2 reacts completely to form PCl3 and PCl5, which are the only products. Determine the mass of PCl3 that forms.