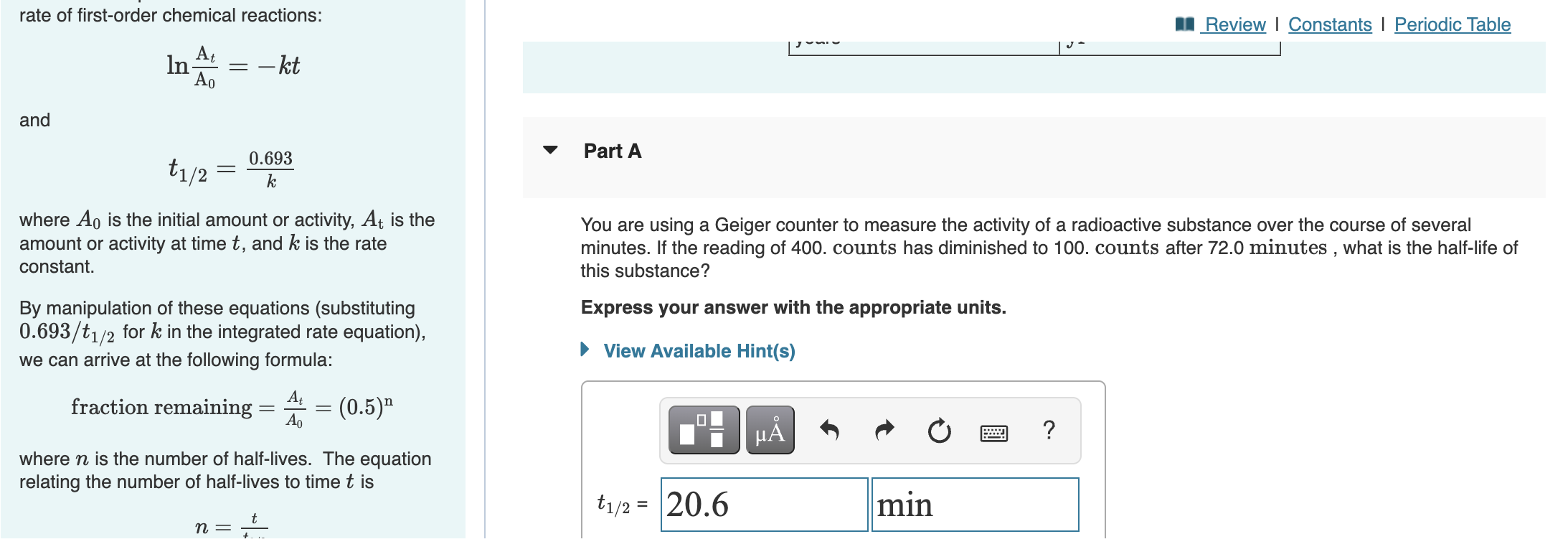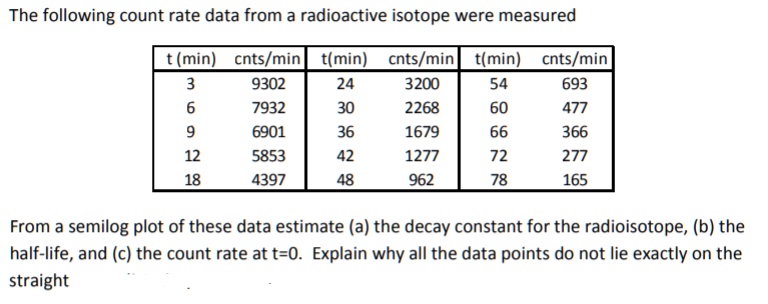
SOLVED: The following count rate data from a radioactive isotope were measured (min) cnts/min t(min) cnts/min t(min) cnts/min 9302 24 3200 54 693 7932 30 2268 60 477 6901 36 1679 66

A first order reaction is found to have a rate constant k= 5.5 xx 10^(-14)s^(-1). Find half-life of the reaction.

Calculate the half life of the first order reaction from their rate constant given as a) 200s^(-) b) 2min^(-1) c) 4 "year"^(-1).

Rate constant of a reaction is 0.0693 min ∧ 1, starting with 10 moles. Rate of reaction after 10 minutes is ???

Define half life of a reaction Derive the relationship between half life and rate constant for a first Order - Chemistry - - 16068871 | Meritnation.com

Chemical Kinetics Class 12 Notes Chemistry Chapter 4 - Learn CBSE | PDF | Reaction Rate | Reaction Rate Constant

A first order reaction is half complete in 45 min. Caluclate the rate constant for the reaction - Find 6 Answers & Solutions | LearnPick Resources

Solve this: Q 5 In a first order reaction, the rate constant is 0 693 hr-1 The half-life - Chemistry - Chemical Kinetics - 12322113 | Meritnation.com

Half life of a first order reaction is 2.1xx10^(12)s. Calculate the rate constant of the reactio... - YouTube

SOLVED:The following data have been obtained for the decomposition of N2 O5(g) at 67^∘ C according to the reaction 2 N2 O5(g) →4 NO2(g)+O2(g) Identify the order of the reaction with respect
✓ Solved: The rate constant for a certain radioactive nuclide is 1.0 × 10^-3 h^-1 .What is the half-life...

The rate constant of a reaction is 0.0693 min^(-1). Starting with 10 mol, the rate of the reaction after 10 min is

The rate constant of a reaction is 0.69 xx 10^(-1) and the initial concentration is 0.2 "mol l"^(-1). The half-life period is

The rate constant of a first order reaction is 0.0693 min^-1 . Time (in minutes) required for reducing an initial concentration of 20mol lit^-1 to 2.5mol lit^-1 is :

The energy of activation and specific rate constant for a first order reaction at 25^∘ C are 100kJ/mole and 3.46 × 10^-5sec^-1 respectively. Determine the temperature at which half - life of
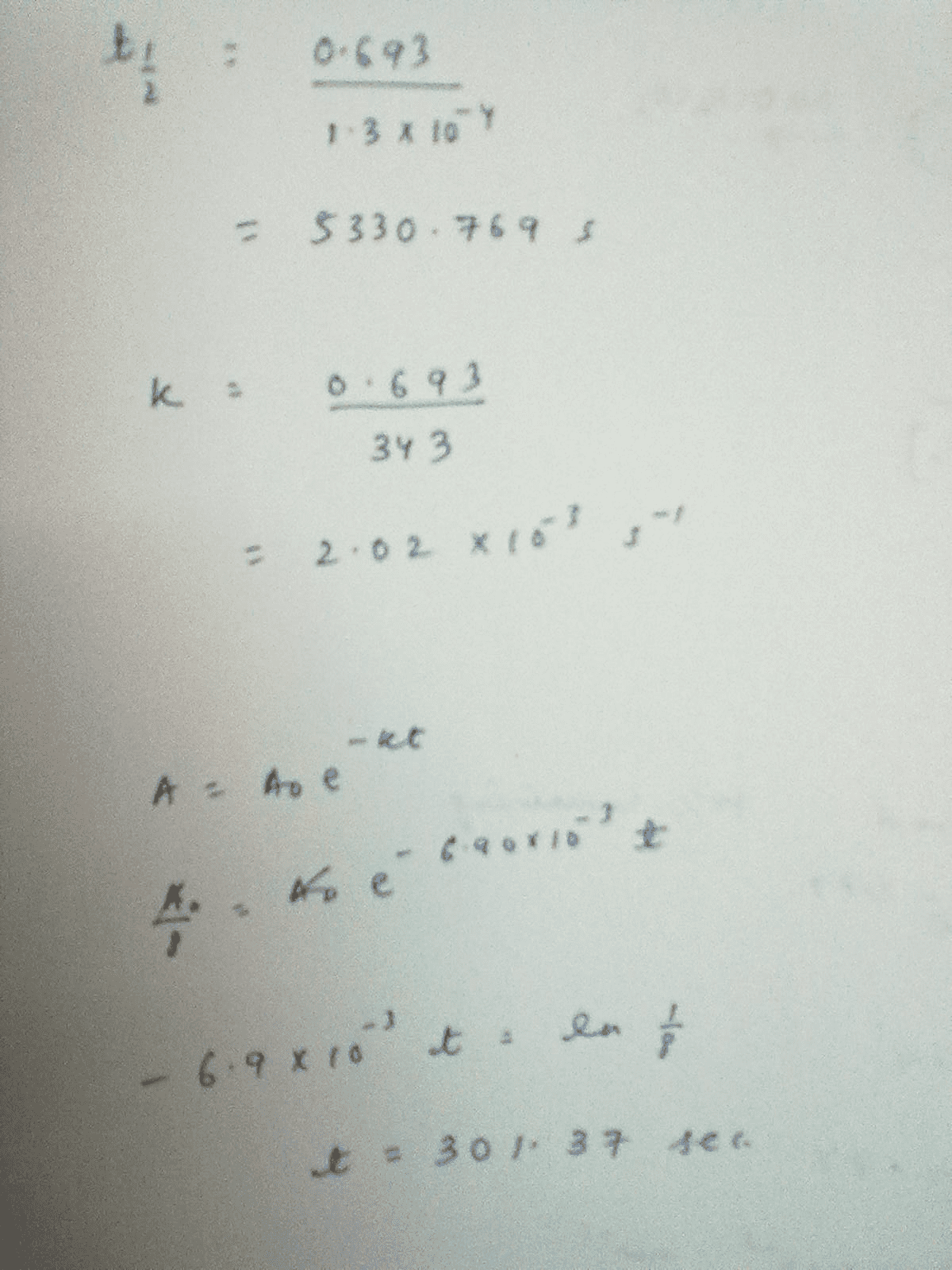
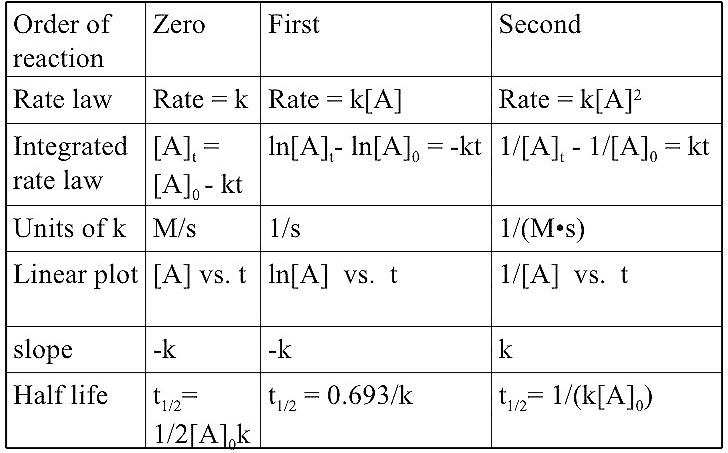


![Solved t1/2 = 1/k[A]. t 0,693/ 1/2 k -2 What is rate the | Chegg.com Solved t1/2 = 1/k[A]. t 0,693/ 1/2 k -2 What is rate the | Chegg.com](https://media.cheggcdn.com/media/560/5609f2c5-63ae-4aef-86f7-fb89896e383e/phpXExk27.png)