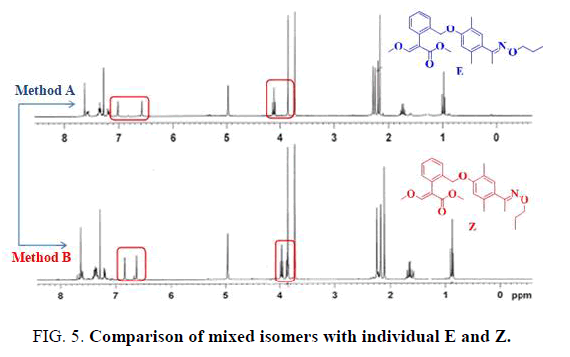
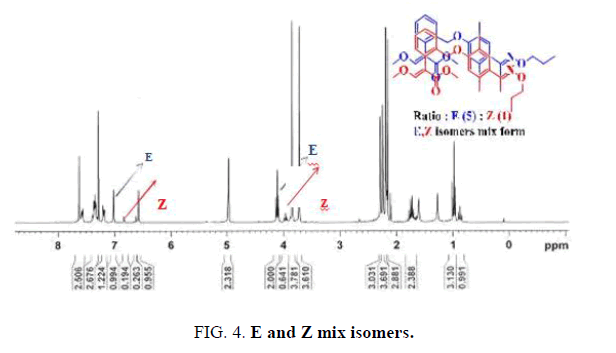
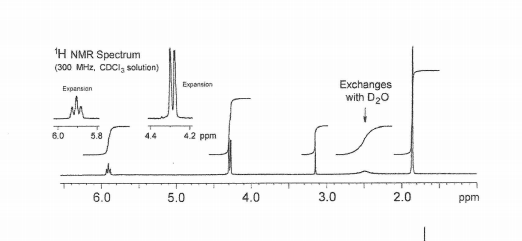
Magnification of a region of the 1 H NMR spectra (400 MHz, CDCl 3 ) for... | Download Scientific Diagram

Can my 13C/1H NMR spectra differentiate between E and Z isomers? I managed to argue that my product is indeed 1-bromo-1,2-diphenylethene, however i cant seem to show that it is the (E)

13 C solid-state NMR spectra of extrudates: nitrofurantoin 3 (E-isomer... | Download Scientific Diagram
A Dynamic NMR Investigation of the Conformational Isomerism in Novel 1,3,4,5-Tetrahydro-2,1-benzoxazepines
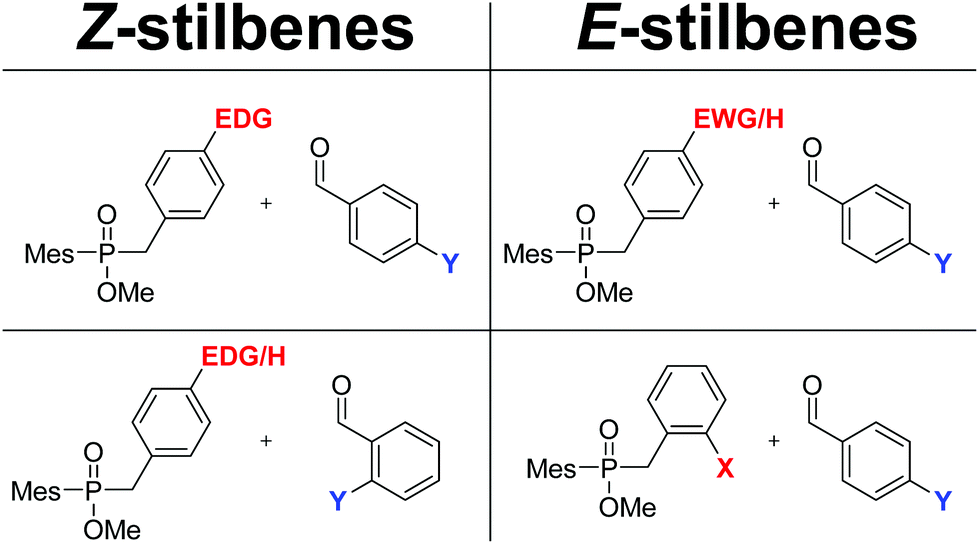
E , Z -Selectivity in the reductive cross-coupling of two benzaldehydes to stilbenes under substrate control - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB01139H
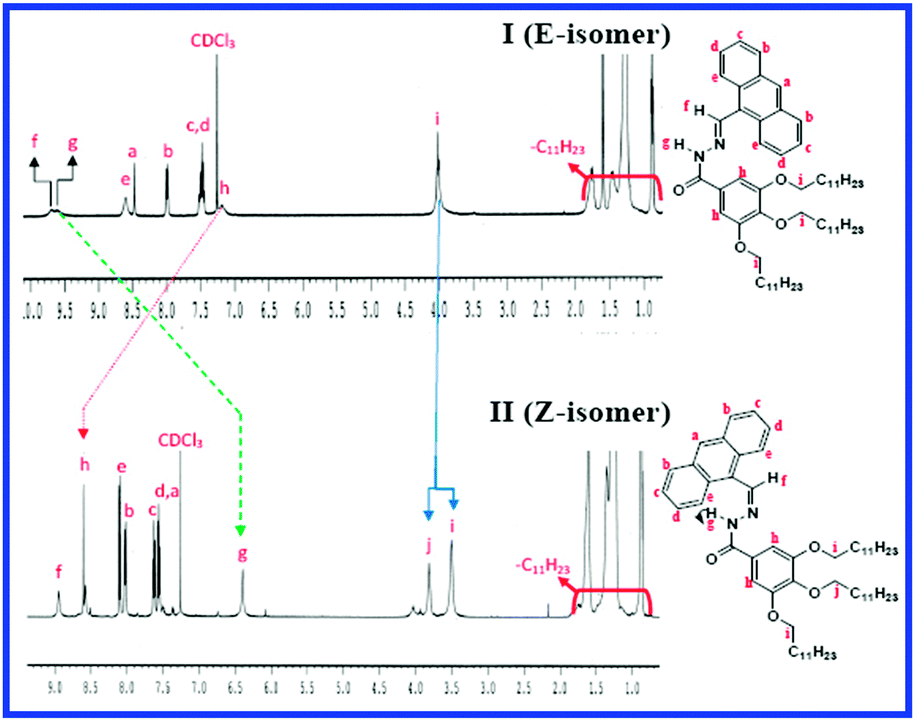
Light induced E – Z isomerization in a multi-responsive organogel: elucidation from 1 H NMR spectroscopy - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC03609G

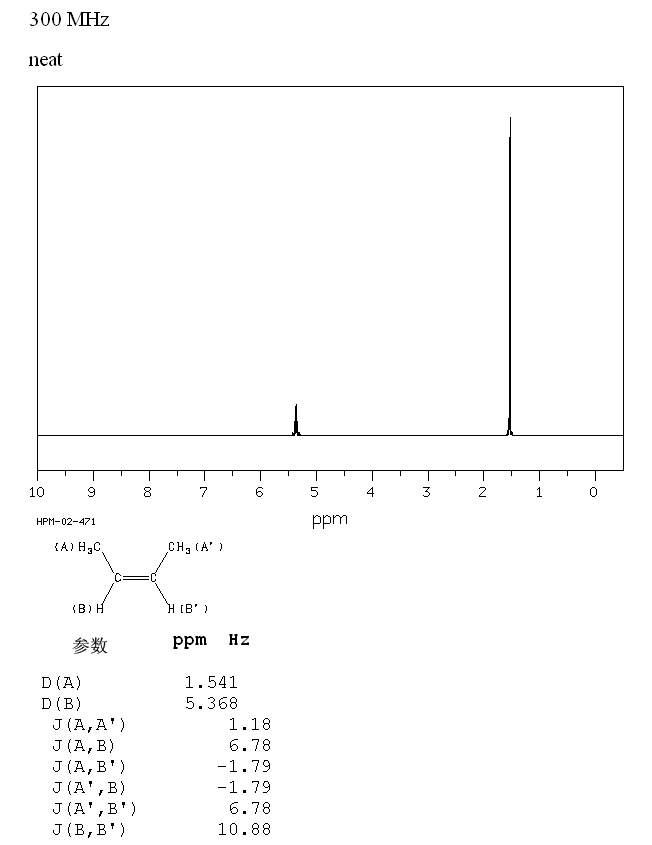
C5H10 E-pent-2-ene 2-pentene Z-pent-2-ene low high resolution H-1 proton nmr spectrum of analysis interpretation of chemical shifts ppm spin spin line splitting H1 cis-pent-2-ene trans-pent-2-ene 1-H nmr doc brown's advanced organic chemistry