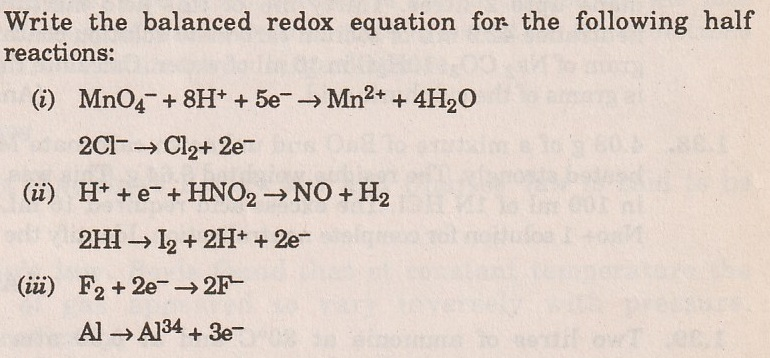The s†an ded reduction potential for the half cell NO3 (aq) + 2H+(aq) + e → NO2 (g) + H2O(l) is 0.78 v . The reduction potnetial in 8M H+ , 1

Understanding the potential band position and e–/h+ separation lifetime for Z-scheme and type-II heterojunction mechanisms for effective micropollutant mineralization: Comparative experimental and DFT studies - ScienceDirect

Radial intensity profiles of H+ and W+ ions from ∼3 to 220 keV/e. Lines... | Download Scientific Diagram