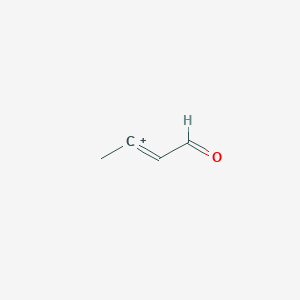
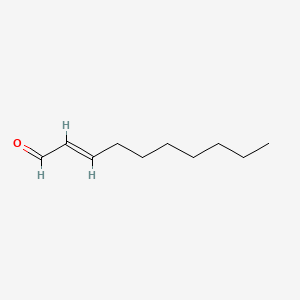
α,β‐Unsaturated Aldehydes as C‐Building Blocks in the Synthesis of Pyridines, 1,4‐Dihydropyridines and 1,2‐Dihydropyridines - Vchislo - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Copper-Mediated Selective Cyanation of Indoles and 2-Phenylpyridines with Ammonium Iodide and DMF | Organic Letters

α,β‐Unsaturated Aldehydes as C‐Building Blocks in the Synthesis of Pyridines, 1,4‐Dihydropyridines and 1,2‐Dihydropyridines - Vchislo - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

BIOACTIVE COMPOUNDS IDENTIFIED IN THE METHANOLIC EXTRACT OF AURICULARIA... | Download Scientific Diagram

Synthesis of Mesoporous Iron Oxides by an Inverse Micelle Method and Their Application in the Degradation of Orange II under Visible Light at Neutral pH | The Journal of Physical Chemistry C
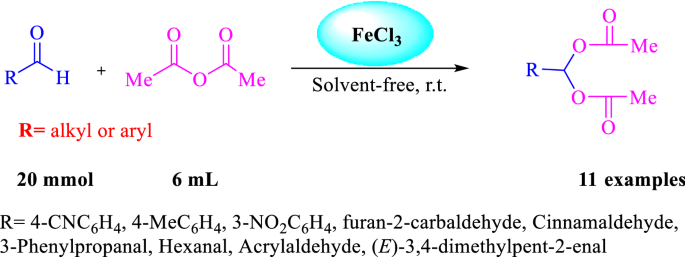
Metal-based Lewis acid catalysts for conversion of a variety of aldehydes with acetic anhydride to gem 1,1-diacetates | SpringerLink

New Chiral Thiophene−Salen Chromium Complexes for the Asymmetric Henry Reaction | The Journal of Organic Chemistry

Chemoselective Ring Closure of 4-(3-Methyl-2-oxo-2,5-dihydro-1H-pyrrol-1-yl)butanal Leading to Pandalizine A | ACS Omega