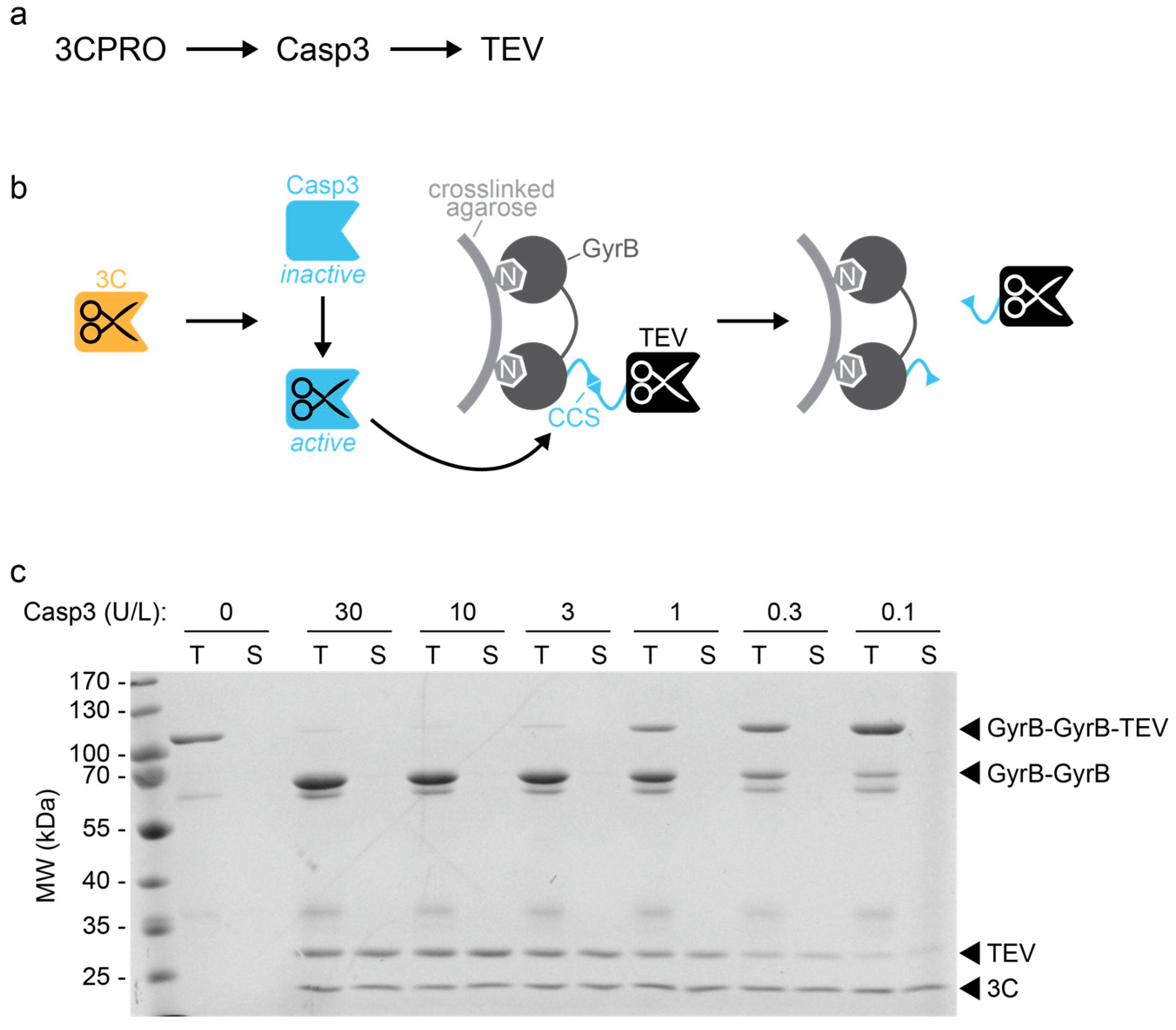
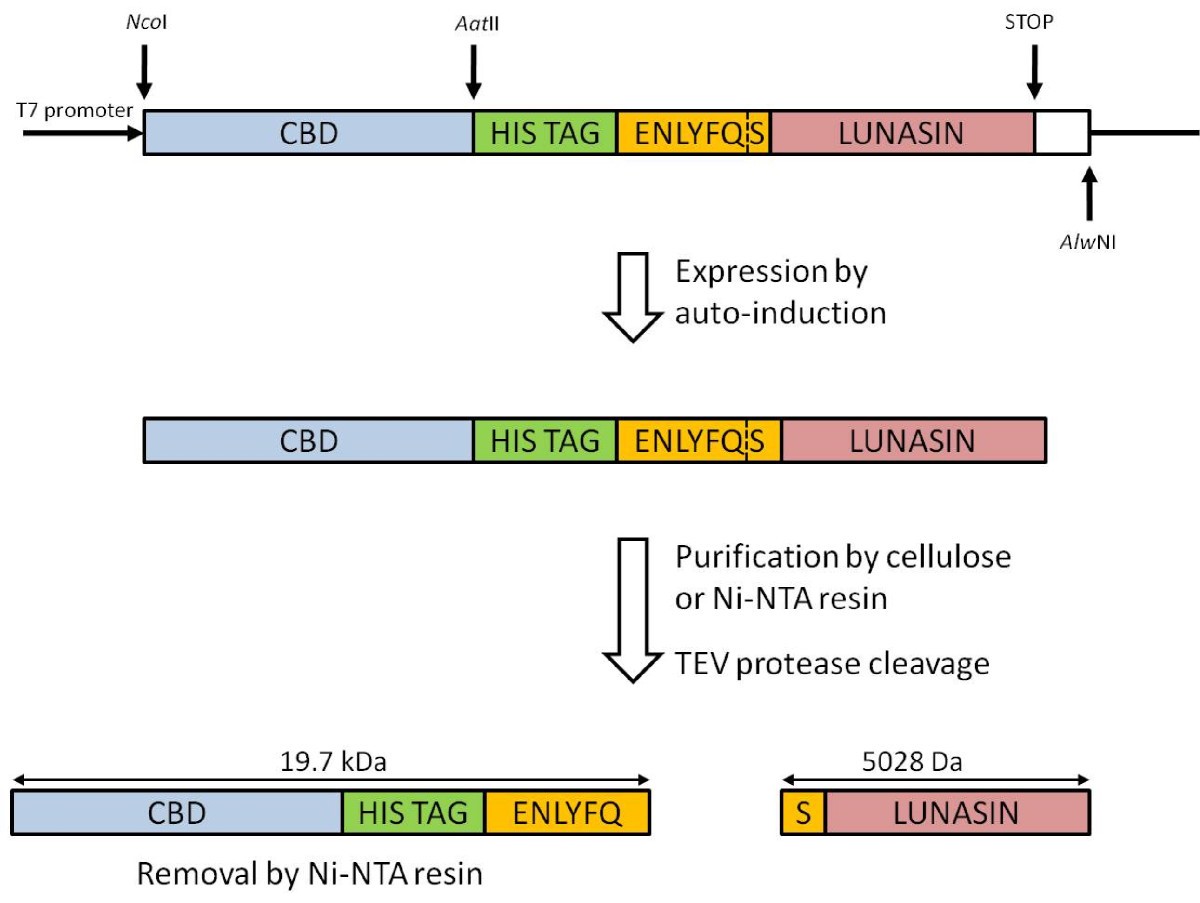
Going native: Complete removal of protein purification affinity tags by simple modification of existing tags and proteases - ScienceDirect

Improved yield, stability, and cleavage reaction of a novel tobacco etch virus protease mutant | SpringerLink
Identification of a Putative Network of Actin-Associated Cytoskeletal Proteins in Glomerular Podocytes Defined by Co-Purified mRNAs | PLOS ONE

Phosphorylation regulates proteolytic efficiency of TEV protease detected by a 5(6)-carboxyfluorescein-pyrene based fluorescent sensor - ScienceDirect
Highly efficient soluble expression, purification and characterization of recombinant Aβ42 from Escherichia coli

Tobacco etch virus (TEV) protease with multiple mutations to improve solubility and reduce self‐cleavage exhibits enhanced enzymatic activity - Nam - 2020 - FEBS Open Bio - Wiley Online Library

TEV protease cleavage of bioGATA-2 bound to streptavidin beads. (A)... | Download Scientific Diagram
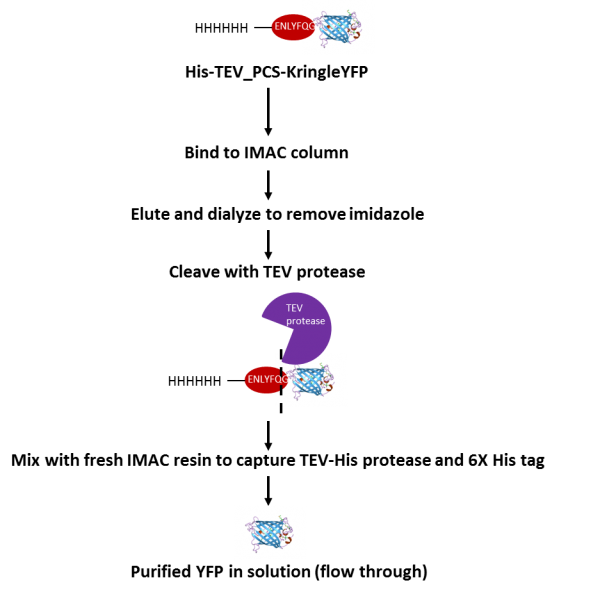
Enzymes - Tobacco Etch Virus (TEV) and Human RhinoVirus (HRV3C) Cysteine Proteases in Vectors | ATUM - ATUM

Tobacco etch virus (TEV) protease with multiple mutations to improve solubility and reduce self‐cleavage exhibits enhanced enzymatic activity - Nam - 2020 - FEBS Open Bio - Wiley Online Library



![PDF] The P1' specificity of tobacco etch virus protease. | Semantic Scholar PDF] The P1' specificity of tobacco etch virus protease. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bb80e47c1b848fe2ae3d1420d56e2f2bb11ade1c/3-Figure1-1.png)




-Western-Blot-NBP2-29909-img0003.jpg)