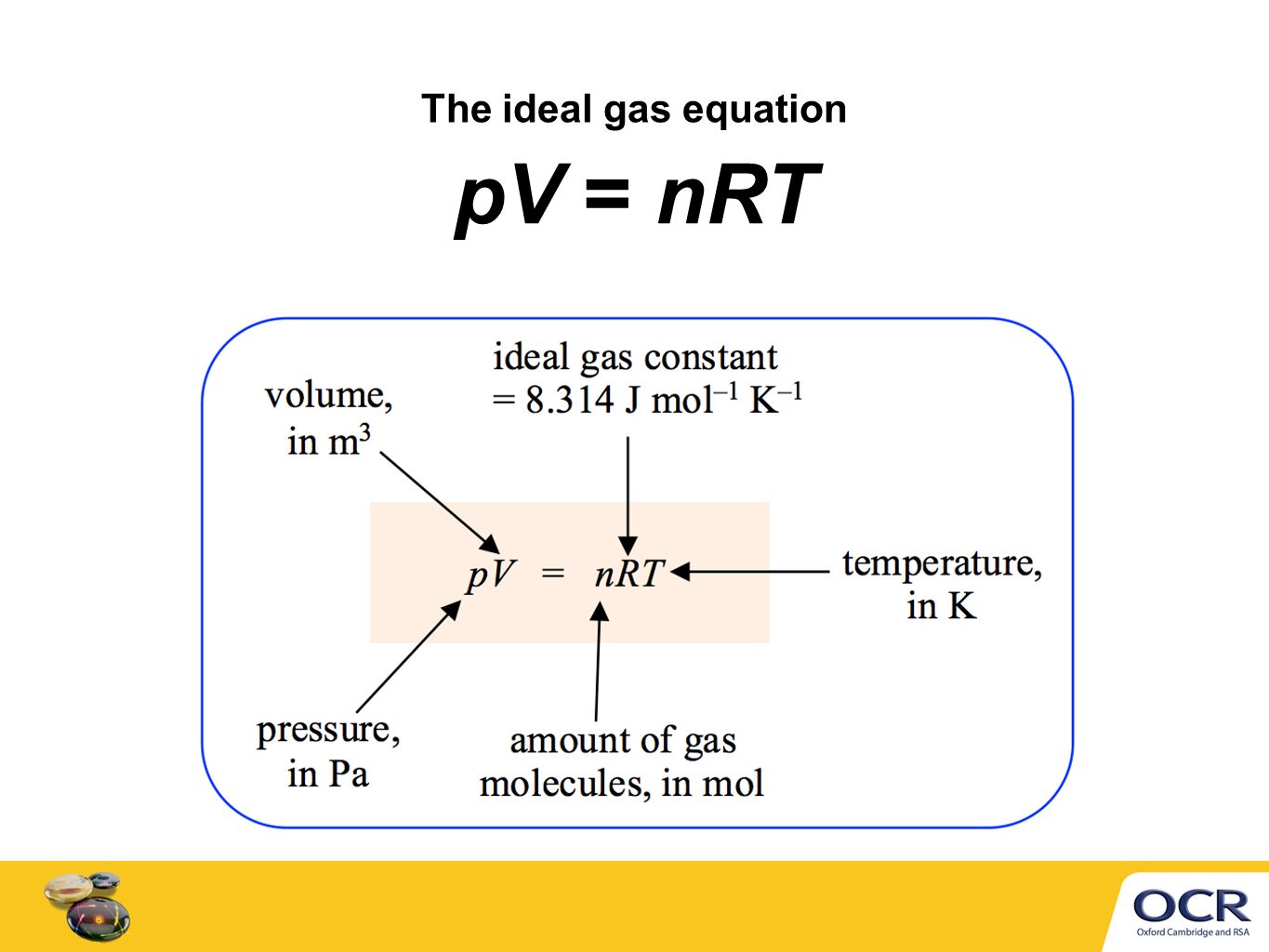
The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download

In Vander Wall's equation (P +(a)/(V^2))(V - b) = RT What are the dimensions of a and b ? Here, P is pressure, V is volume, T is temperature and R is
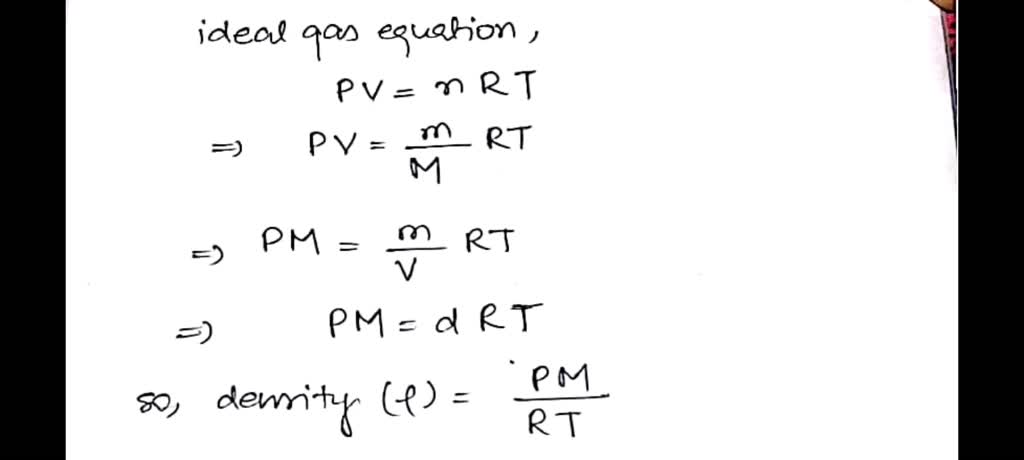
SOLVED: How can one determine the density of a gas from the ideal gas law equation? m By definition, P = m/V where P = density ; m = mass; V=volume.
p+a/v)(v b) =RT, p=pressure,v=volume,R,a,b are constant, T=absolute temperature. find dimension of a/b^2

Ideal Gas Law PV = nRT re-arrange n V = P RT n = molar mass (g/mol) mol gas= mass gas (g) mass of sample V x molar mass = P RT =

Ideal Gas Law PV = nRT re-arrange n V = P RT n = molar mass (g/mol) mol gas= mass gas (g) mass of sample V x molar mass = P RT =

Ideal Gas Law PV = nRT re-arrange n V = P RT n = molar mass (g/mol) mol gas= mass gas (g) mass of sample V x molar mass = P RT =

Ideal-Gas Equation The constant of proportionality is known as R, the gas constant. © 2009, Prentice-Hall, Inc. - ppt download

Ideal Gas Law PV = nRT re-arrange n V = P RT n = molar mass (g/mol) mol gas= mass gas (g) mass of sample V x molar mass = P RT =

SOLVED: The van der Waals equation is: P + Wo) = b RT. Where; P = pressure (atm) n = number of moles (mol) R = ideal gas constant V = volume (















