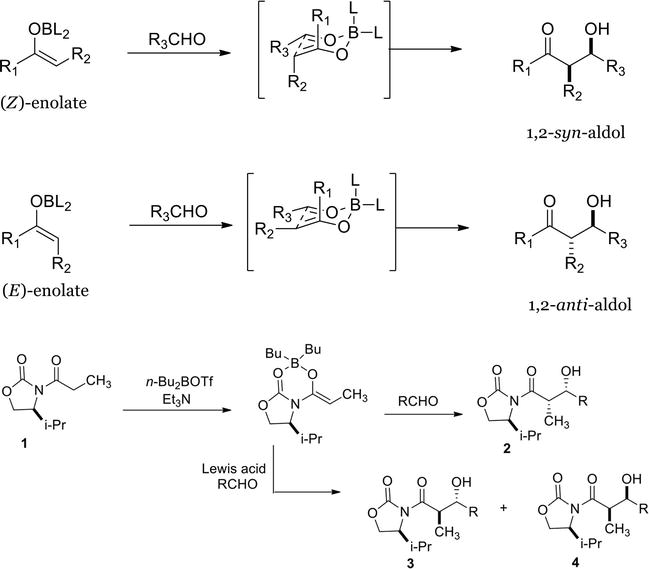
Transition Metal Catalyzed Insertion Reactions with Donor/Donor Carbenes - Bergstrom - 2021 - Angewandte Chemie International Edition - Wiley Online Library
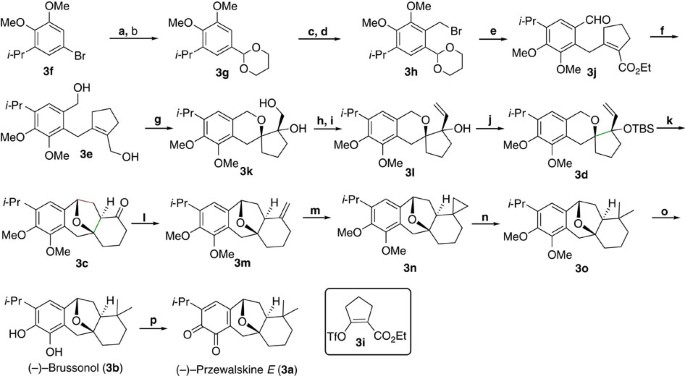
Tandem C–H oxidation/cyclization/rearrangement and its application to asymmetric syntheses of (−)-brussonol and (−)-przewalskine E | Nature Communications

Connecting and Analyzing Enantioselective Bifunctional Hydrogen Bond Donor Catalysis Using Data Science Tools | Journal of the American Chemical Society

In situ generation of N-unsubstituted imines from alkyl azides and their applications for imine transfer via copper catalysis | Science Advances

Stereoinduction in Metallaphotoredox Catalysis - Lipp - 2021 - Angewandte Chemie International Edition - Wiley Online Library
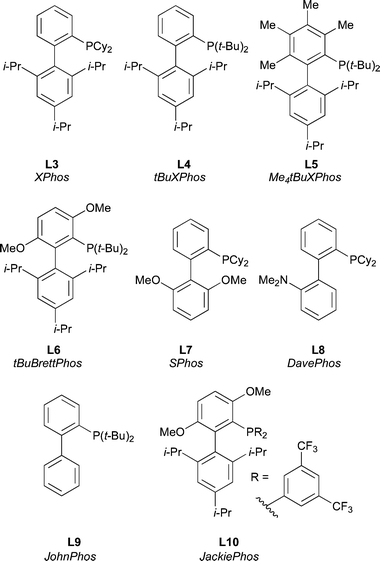
Dialkylbiaryl phosphines in Pd-catalyzed amination : a user's guide - Chemical Science (RSC Publishing) DOI:10.1039/C0SC00331J
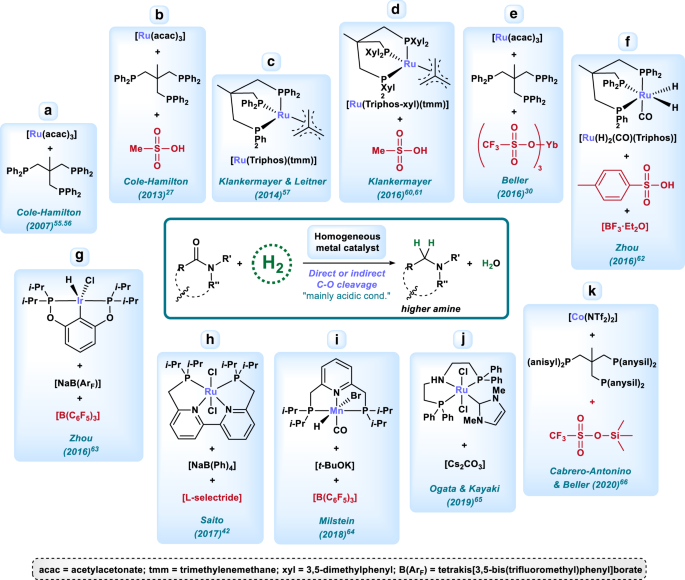
Homogeneous and heterogeneous catalytic reduction of amides and related compounds using molecular hydrogen | Nature Communications

Synthesis and Reactivity of 1,1‐Diborylalkanes towards C–C Bond Formation and Related Mechanisms - Miralles - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library
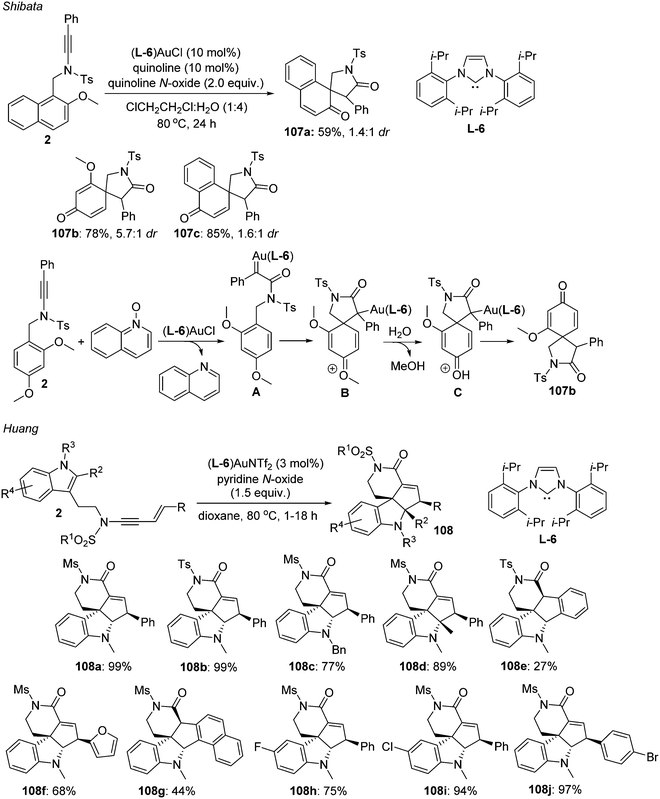
Asymmetric synthesis with ynamides: unique reaction control, chemical diversity and applications - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D0CS00769B
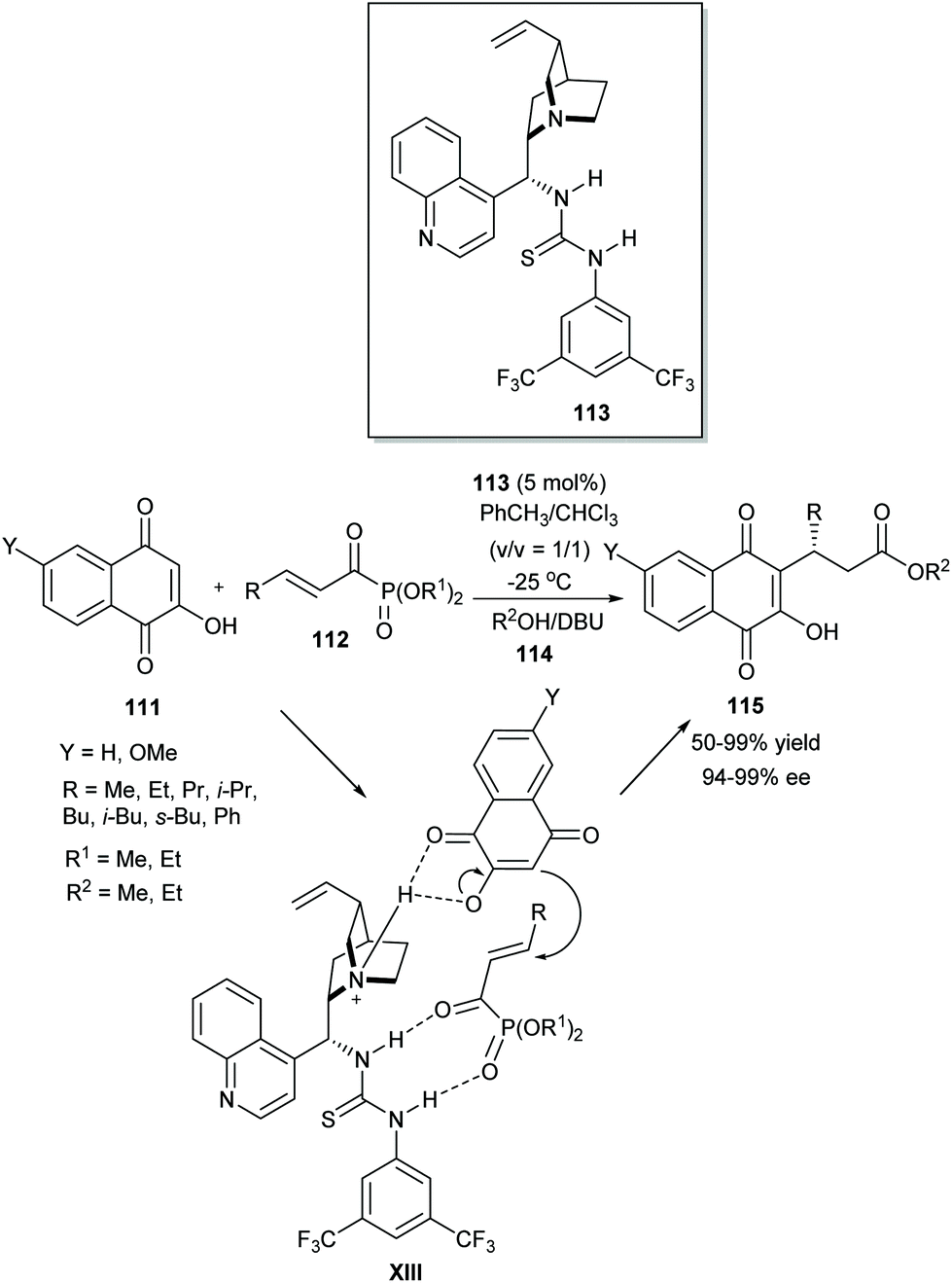
Recent applications of thiourea-based organocatalysts in asymmetric multicomponent reactions (AMCRs) - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB00595A

Lithium Diisopropylamide-Mediated Enolization: Catalysis by Hemilabile Ligands | Journal of the American Chemical Society

Dinuclear Nickel Complexes in Five States of Oxidation Using a Redox-Active Ligand | Inorganic Chemistry
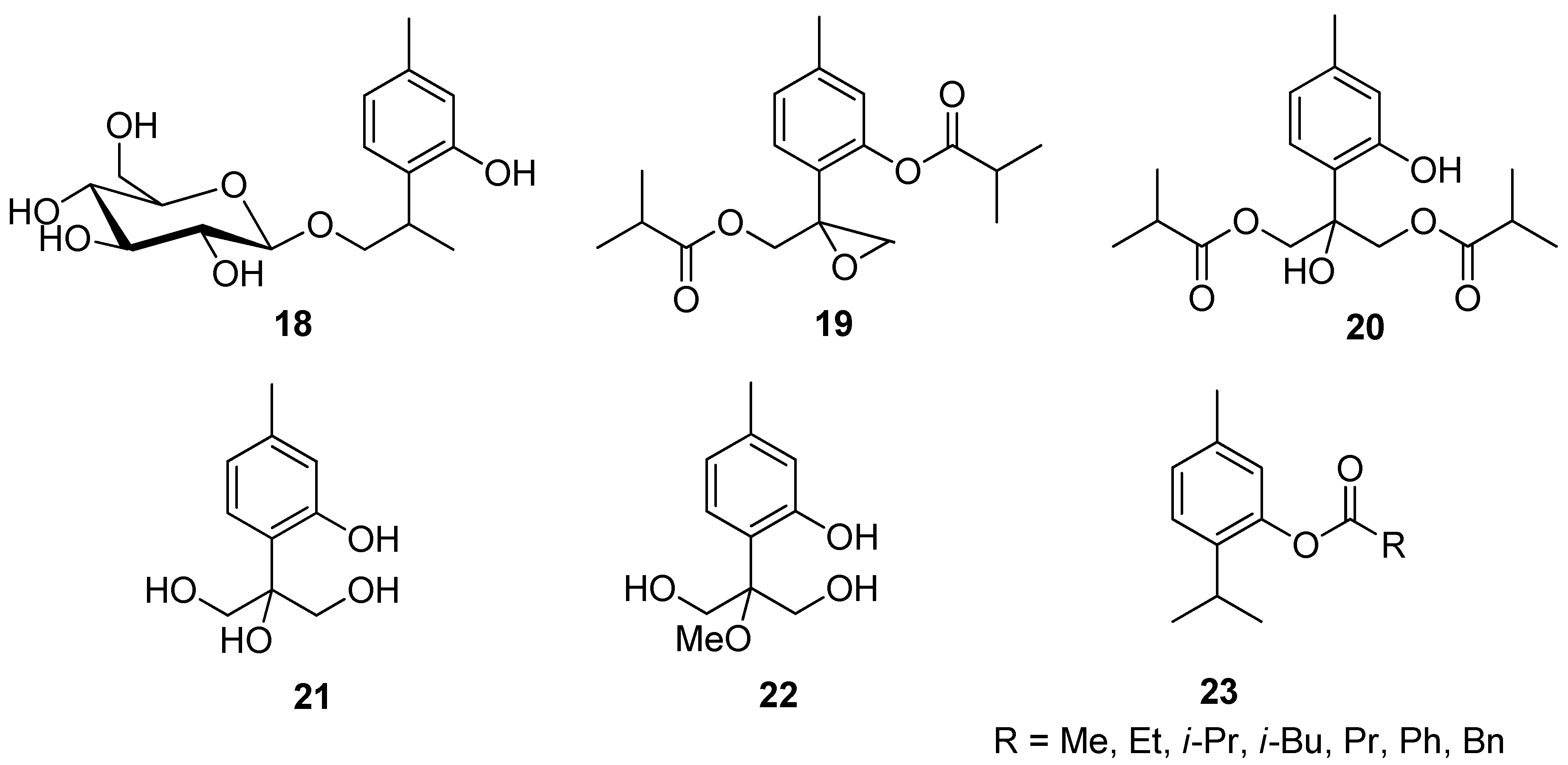
IJMS | Free Full-Text | Monoterpenes and Their Derivatives—Recent Development in Biological and Medical Applications