The most probable mechanism of Reaction c, PA-O2 + O2H → PA-O2H + O2,... | Download Scientific Diagram

Pa O2 and Pa CO2 change over time during apnoea. Change in arterial... | Download Scientific Diagram

Henry\'s law constant for oxygen and nitrogen dissolved in water at 298 K are `2.0 xx 10^(9)` Pa... - YouTube

Natalie Pinkham attending the National Television Awards 2019 held at the O2 Arena, London. PRESS ASSOCIATION PHOTO. Picture date: Tuesday January 22, 2019. See PA story SHOWBIZ NTAs. Photo credit should read:
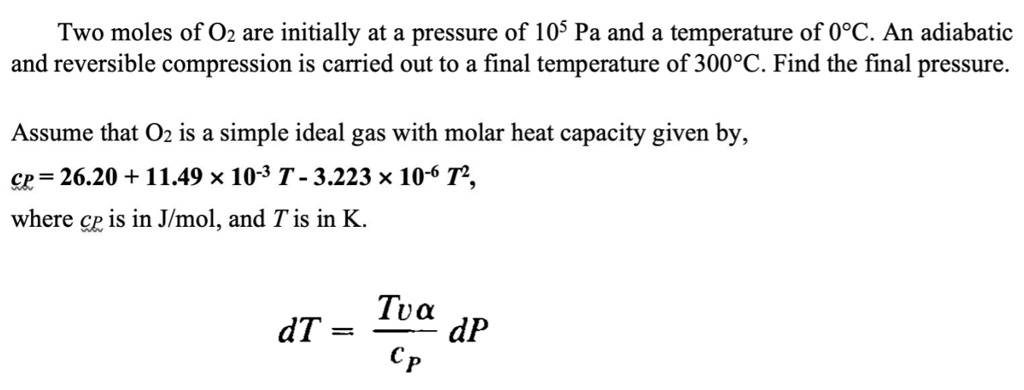
SOLVED: Two moles of O2 are initially at a pressure of 105 Pa and a temperature of 0C. An adiabatic and reversible compression is carried out to a final temperature of 300C.

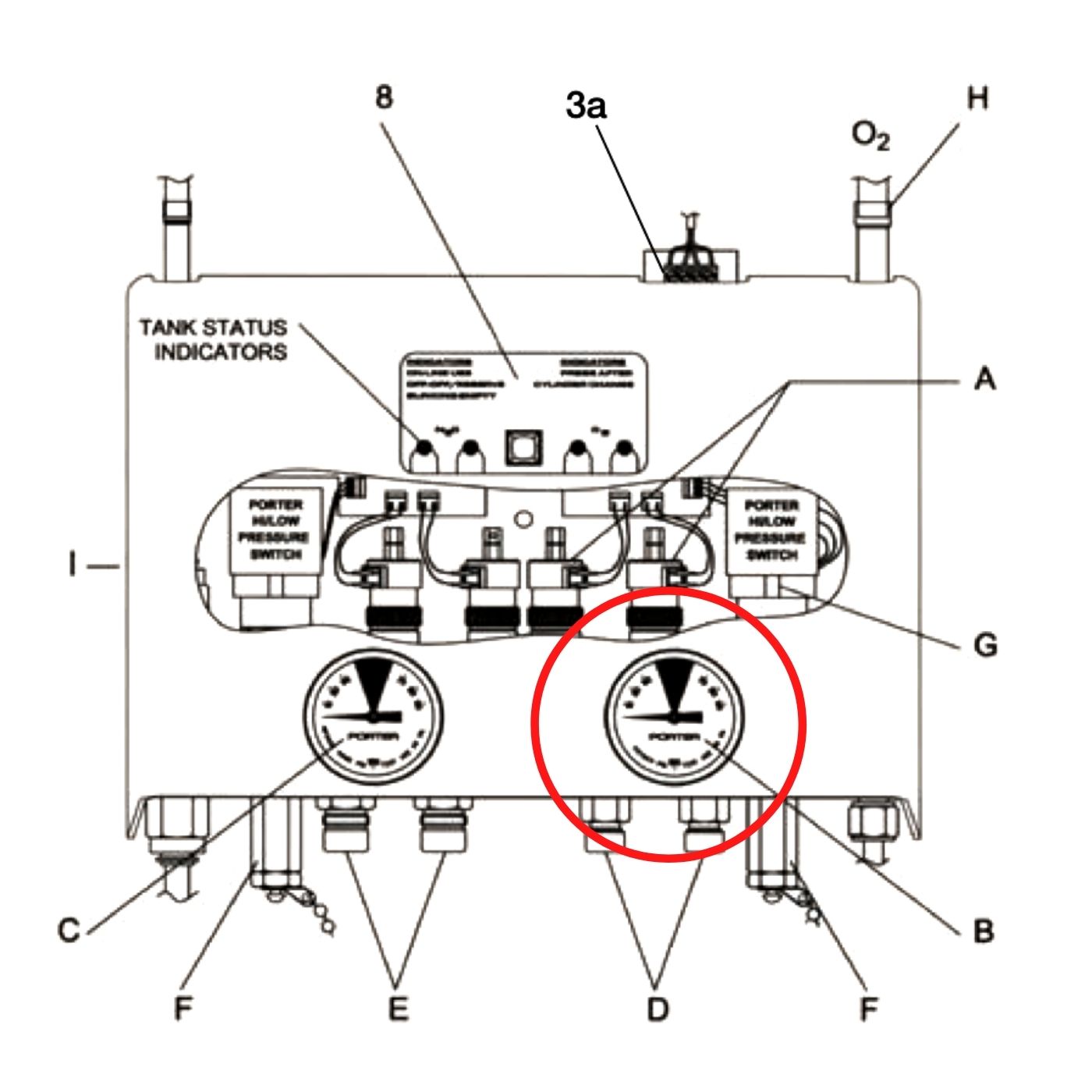
ZSFG AM Report 8/21/2017: Oxygen Delivery Systems and Use of PA Catheters in Undifferentiated Shock | UCSF Internal Medicine Chief Resident Hub

The Henry's law constant for the solubility of oxygen in water is 3.3 × 10^ -4 M/atm at 12 ^o C. Air is 21 mol

SOLVED: Determine the diffusivity of oxygen O2 in the gas mixture at 250 K and 1 atm. The operating conditions are as foll CO10%:Do2-co2=1.78x10-5m/Sec CO16%:Doz-co=1.99x10~5m/Sec N50%Do2-N=1.72x10-5m/Sec O24% Pa tor estion Select one: