New Phytologist Supporting Information Methane Production and Emissions in Trees and Forests Kristofer R. Covey and J. Patrick M

OpenStax College Physics Solution, Chapter 23, Problem 36 (Problems & Exercises) | OpenStax College Physics Answers
A unit cell of sodium chloride has four formula units.The edge length of unit cell is 0.6 nm.What is the density of sodium chloride? Kindly provide a solution with clear cut step
✓ Solved: Nickel has a face-centered cubic unit cell. The density of nickel is 6.84g∕cm^3. Calculate...
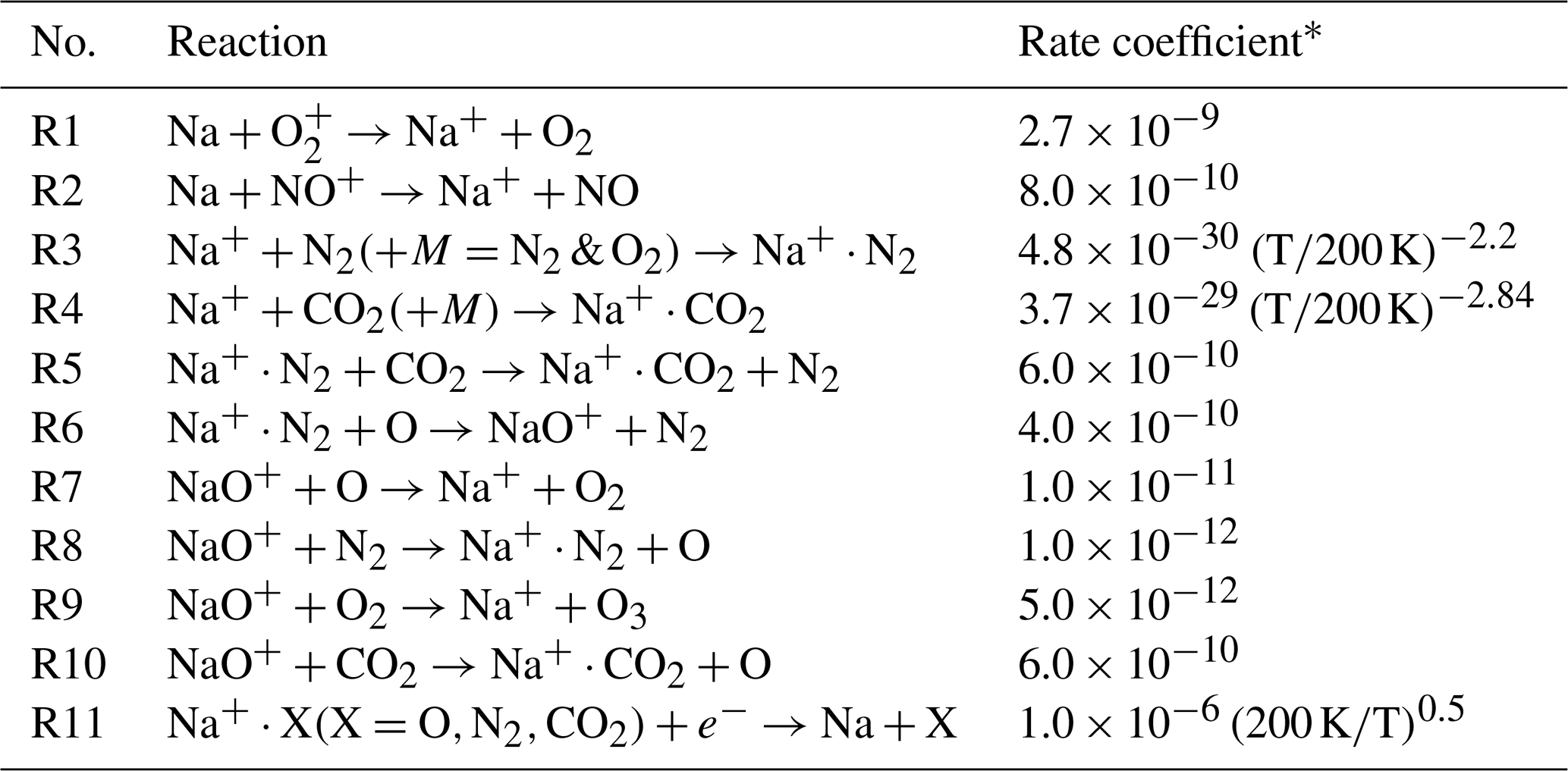
ACP - The sporadic sodium layer: a possible tracer for the conjunction between the upper and lower atmospheres

Molybdenum forms body-centred cubic crystals whose density is `10.3 g cm^(-3)` . Calculate the edge - YouTube

Metal has an fcc lattice. The edge length of the unit cell is 404 pm. The density of the metal is 2.72 g cm ^-3 . The molar mass (in g mol ^-
What is the distance between Na+ and Cl– ions in NaCl crystal if its density is 2.165 g cm–3 ? [Atomic Mass of Na = 23u, Cl = 35.5 u; Avogadro's number = 6.023 × 1023]

If it takes 38.70cm³ of 1.90 M NaOH to neutralize 10.30cm³ of H2SO4 in a battery, What's the Molarity of - Brainly.com
A solenoid coil has 50 turns per cm along its lenght and a cross sectional area of 4cm^2. Now 200 turns of another wire are wounded round the first solenoid coaxially,the coils

SOLVED:II Two long, parallel transmission lines 40.0 cm apart carry 25.0 A and 75.0 A currents. Find all locations where the net magnetic field of the two wires is zero if these