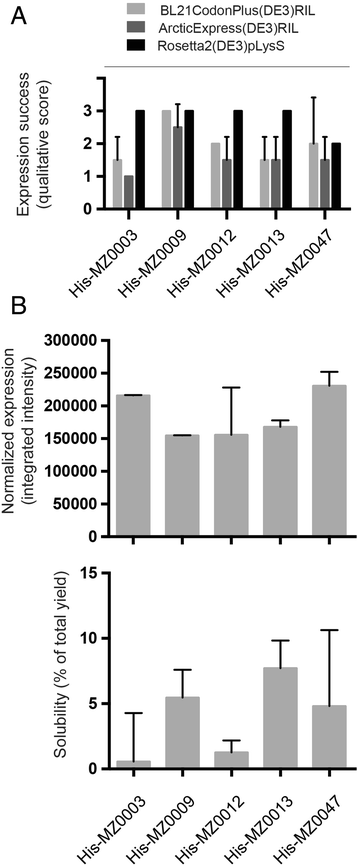
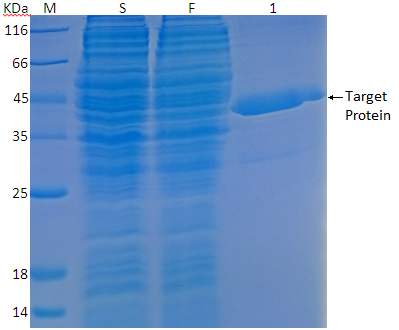
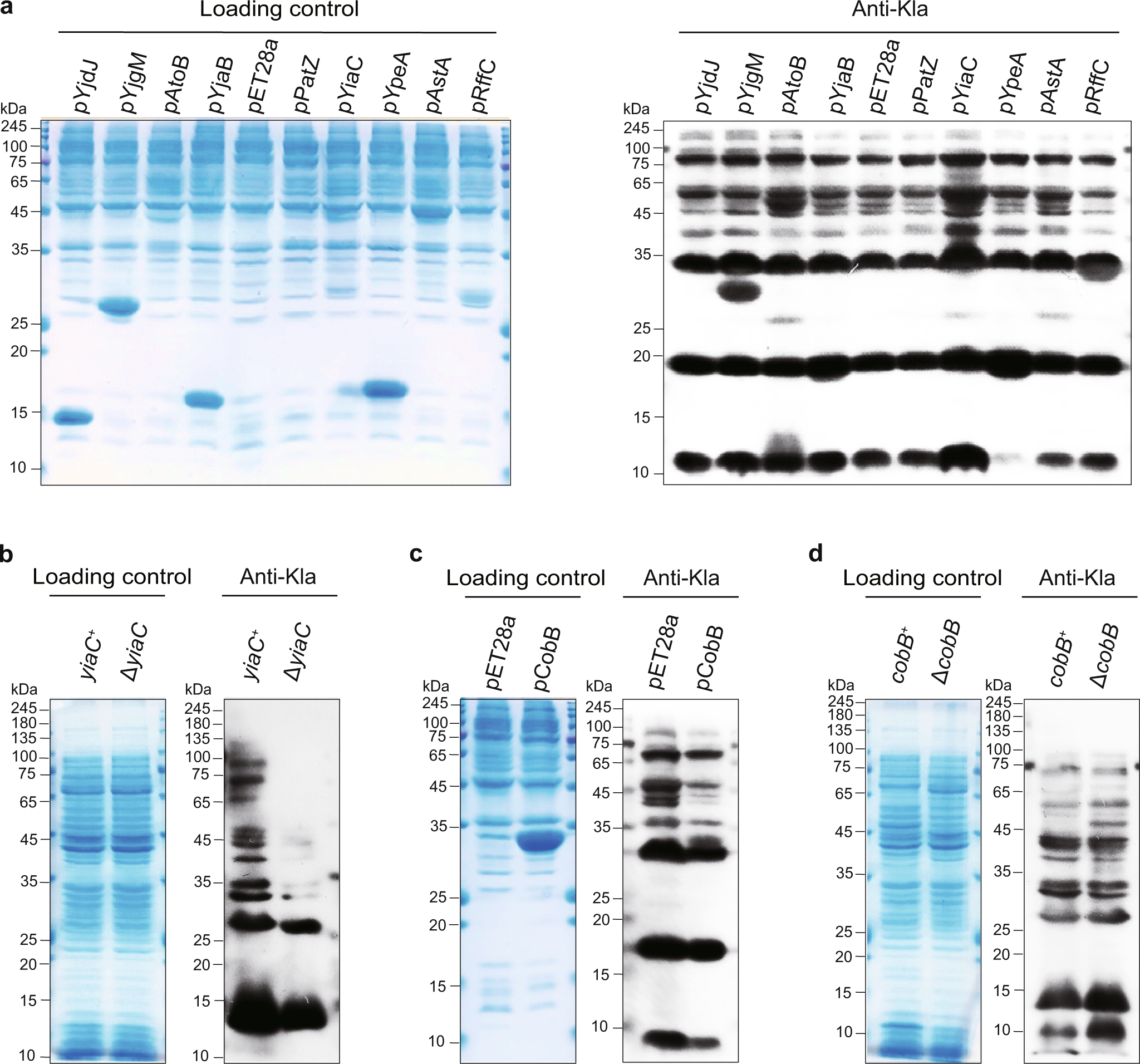
Safety Aspect of Recombinant Protein Produced by Escherichia coli: Toxin Evaluation with Strain and Genomic Approach | IntechOpen

A novel method for removing contaminant Hsp70 molecular chaperones from recombinant proteins - Morales - 2019 - Protein Science - Wiley Online Library
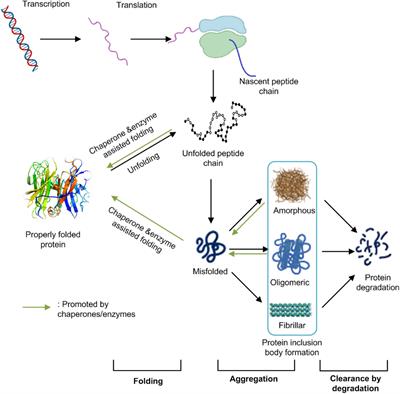
Frontiers | Challenges Associated With the Formation of Recombinant Protein Inclusion Bodies in Escherichia coli and Strategies to Address Them for Industrial Applications
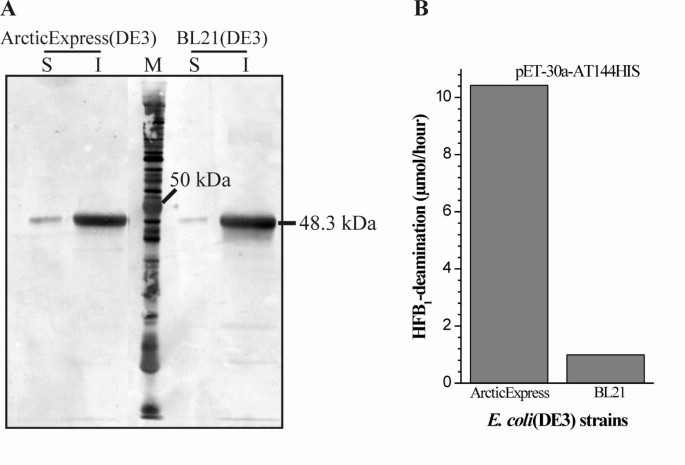
Enhancement of solubility in Escherichia coli and purification of an aminotransferase from Sphingopyxis sp. MTA144 for deamination of hydrolyzed fumonisin B1 | Microbial Cell Factories | Full Text
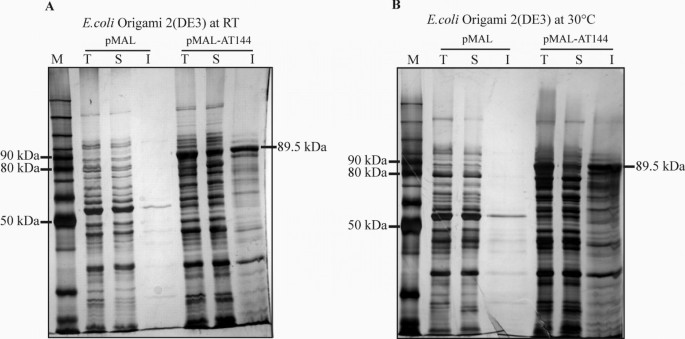
Enhancement of solubility in Escherichia coli and purification of an aminotransferase from Sphingopyxis sp. MTA144 for deamination of hydrolyzed fumonisin B1 | Microbial Cell Factories | Full Text

Expression, characterization, and site-specific covalent immobilization of an L-amino acid oxidase from the fungus Hebeloma cylindrosporum | SpringerLink

A fast and simple method to eliminate Cpn60 from functional recombinant proteins produced by E. coli Arctic Express - ScienceDirect
Design, Construction, and Expression of Autoactivated Thrombin Precursor Gene in Escherichia coli Arctic Express (DE3) Research
Biochemical Characterization of Putative Adenylate Dimethylallyltransferase and Cytokinin Dehydrogenase from Nostoc sp. PCC 7120 | PLOS ONE